In the global pursuit of net-zero carbon emissions, countries around the world are actively seeking sustainable alternative energy solutions, and lithium-ion batteries have become a core power source for electric vehicles and energy storage systems. Professor Jeng-Han Wang of the Department of Chemistry at National Taiwan Normal University and his research team have integrated density functional theory (DFT) with spectroscopic experiments to investigate the lithium-storage mechanism of shear-phase H-Nb₂O₅, a promising anodic material for lithium-ion batteries, e in the development of fast-charging lithium-storage materials.
Professor Wang specializes in applying DFT calculations and simulations to elucidate catalytic mechanisms, with a focus on understanding the intrinsic properties of advanced functional materials for applications in rechargeable batteries, fuel cells, electrochemical devices, and spintronics. This study originated from his postdoctoral research at the School of Materials Science and Engineering at the Georgia Institute of Technology.
Speaking about how the project began, Wang described it as “a lucky coincidence.” During his postdoctoral years, he built close collaborative relationships with faculty in Georgia Tech’s materials science department, and those collaborations have persisted after his return to Taiwan. He initially worked on fuel cells, but as he saw the rise of electric vehicles and lithium-ion batteries, he began to consider whether ceramic materials could also be applied in this area.
“If we imagine the cathode and anode of a lithium-ion battery as two hotels, then the lithium ions move back and forth between them during charging and discharging like guests.” The central material in this study, H-Nb₂O₅, has a Wadsley-Roth shear structure. Its lattice arrangement is particularly well suited to the reversible insertion and accommodation of lithium ions. It can stably host lithium-ion transport without causing significant structural degradation, while also offering good electronic conductivity, making it an ideal material for high-rate batteries capable of rapid charging and discharging.
In this study, Wang’s team used DFT calculations to simulate in depth the process from Li₀.₁Nb₂O₅ to Li₁.₀Nb₂O₅, revealing changes in electronic distribution and structural response as lithium ions enter the lattice. The research showed that the initially adsorbed lithium ions release electrons into Nb d orbitals, including dxy, dyz, and dzx, gradually transforming the material from an insulating to a conductive state. After lithium adsorption, the band gap narrows significantly, indicating that the material has acquired electronic conductivity.
In addition to developing a new class of H-Nb₂O₅-based materials, the study also employed an alternative and more accessible experimental strategy. Traditionally, investigations of lithium-ion behavior in materials often rely on expensive synchrotron radiation or nuclear magnetic resonance instruments. This work instead chose the more accessible route of Raman spectroscopy, which not only lowers experimental costs but also offers a more widely available analytical pathway. Wang noted, however, that Raman spectra contain many signals, making them less precise and harder to analyze. By simulating the vibrational density of states, or VDOS, the team was able to disentangle these complex signals and successfully map them onto specific bonds and vibrational modes in the structure.
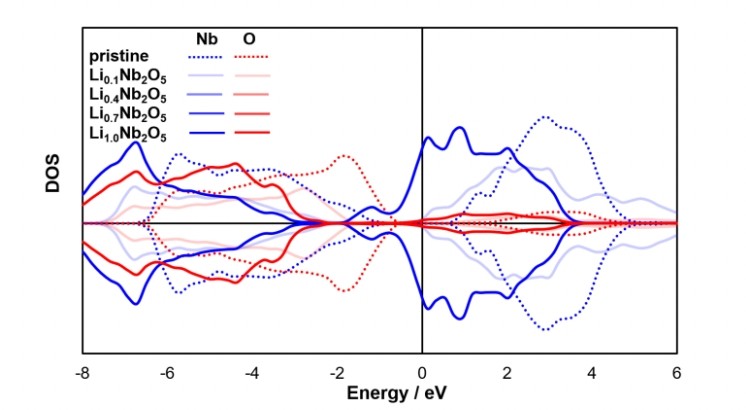
Figure caption: Density-of-states (DOS) analysis of pristine and lithiated H-Nb₂O₅. The Nb d band and O p band are plotted in blue and red, respectively. Pristine H-Nb₂O₅ is shown with dotted lines. Solid lines represent lithiated H-Nb₂O₅, with darker curves indicating higher levels of lithium adsorption.
Lithium-ion technologies have a wide range of applications and remain a highly active area of research. Wang noted that lithium-ion systems are found everywhere in daily life, from mobile phones and computers to cars and scooters. At the university level, however, this work still belongs to the realm of basic research. Materials development also proceeds through continual trial and error: researchers discover new systems and materials through repeated attempts, then return to the literature to identify ways of developing new technologies and materials.
Discussing possible next steps, Wang said that this study is still in the basic research stage, and future work could further extend niobium oxide materials in new directions. For example, lithium atoms could be replaced with other elements in an effort to modify their interaction with lithium ions. Or, when lattice defects appear, it may be necessary to adjust the stoichiometry, altering the composition and structure of the material to create properties tailored to researchers’ goals.
(This article was provided by The Center of Public Affairs.)
Source: Li, T., Huang, S., Kane, N., Wang, J. H., Luo, Z., Zhang, W., Nam, G., Zhao, B., Qi, Y., & Liu, M. (2023). Operando Raman and DFT Analysis of (De)lithiation in Fast-Charging, Shear-Phase H-Nb₂O₅. ACS Energy Letters, 8(7), 3131-3140. https://doi.org/10.1021/acsenergylett.3c01031



